Tag: polyQ
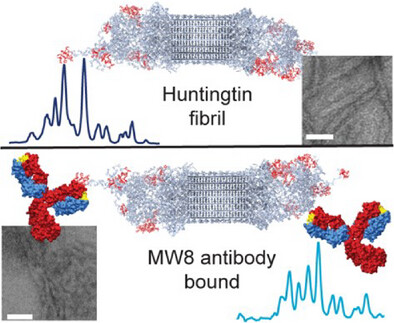
Publication: how antibodies bind to the fuzzy coat of Huntingtin protein fibrils.
Congratulations to Raffaella for the publication of her second first-author paper, this time on the use of NMR spectroscopy to study how antibodies bind to the aggregated state of huntingtin protein fragments, relevant to Huntington’s disease. This work is part of our ongoing research on the biochemistry of Huntington’s disease, and was supported by funds from the European Huntington Disease Network EHDN and the CampagneTeam Huntington. Antibodies are widely used as diagnostic tools in HD research, and also are considered as potential treatment modalities. An example of the latter approach can be found in the case of Alzheimer’s disease, where […]

Publication: Impact of aggregation inhibitors on protein aggregation and aggregate toxicity.
Congratulations to Greeshma Jain, Marina Trombetta-Lima and the whole team of co-authors for the publication of our most recent paper on Huntington’s disease (HD), in the journal Nature Communications. This paper is actually the fourteenth peer-reviewed publication from the lab on HD research, since our first paper in 2011. It was enabled by crucial funding from the CampagneTeam Huntington foundation, who supported our HD research program when the Van der Wel group moved back to the Netherlands. In this new report we describe our use of structural analysis (NMR and EM) to look how aggregation-inhibiting small molecules (polyphenols) change the […]

Publication: New paper about photochemical approaches to studying polyQ protein aggregation.
Congratulations to PhD student Raffaella Parlato, Dr. Jana Volaric, and their collaborators, on the publication of a nice new paper in the Journal of the American Chemical Society. This is the final result of an idea from some years ago, which came together very nicely thanks to a great team of collaborators. The goal of this work was to explore the idea of putting polyglutamine aggregation under some degree of photo-control. In prior work, it has been shown that b-hairpin formation is a key step in the aggregation process of expanded polyQ proteins. Azobenzene-based groups can be used to favor […]

Publication: Review article on polyglutamine protein analysis by solid-state NMR spectroscopy.
Our new review article summarizing progress in polyglutamine protein studies by solid-state NMR is now available on the website of the journal Biochemical Society Transactions. This is an invited mini-review that was requested to cover the progress made in our understanding the aggregation and misfolding of mutant polyglutamine proteins implicated in diseases such as Huntington’s disease and several versions of spinocerebellar ataxia (SCA). We describe how studies from ssNMR has been used to better understand the structure of these protein aggregates, and thus helps us analyze how the aggregation process occurs and how aggregates may play a role in these […]

Publication: Dynamics-based spectral editing to see (fiber) surfaces by solid-state NMR.
Congratulations to Dr. Irina Matlahov and (alum) Dr. Jennifer Boatz on the publication of their new paper in the Journal of Structural Biology X. The paper is entitled “Selective observation of semi-rigid non-core residues in dynamically complex mutant huntingtin protein fibrils“. It describes our latest research on the misfolded protein deposits associated with Huntington’s disease (HD), specifically looking at what happens on the surface of these protein fibrils. In previous work we have studied the structure of these nanometer-sized fibrils formed by mutant huntingtin’s exon 1 fragment, using ssNMR, EM and other methods. In our earlier studies we used dynamics-sensitive […]
News: Dutch-language radio
Recently our research was covered (briefly) on a Dutch radio station, featuring an interview with Patrick. It is mostly about our work applying ssNMR to studying how proteins aggregate in Huntington’s disease. You can find the recording online at the NPO radio website. More information about our Huntington disease research can also be found on this page, and in two recent webinars.

Upcoming event: BPS networking event on polyQ biophysics
Hereby an early announcement of an upcoming online event hosted by our group and the group of Markus Miettinen: “Biophysics of polyglutamine aggregation: how does it start and how does it end?“ This is an online event is organized in context of the networking event series sponsored and organized by the Biophysical Society (BPS). Our event aims to bring together international scientists working on the biophysics of polyglutamine (polyQ) protein aggregation. The event will take place on July 6th 2022, from 15:00-18:00 CET. More information can be found at the page from the BPS or our own institute. An online […]

Webinar: Structural biology of Huntington’s disease.
On May 15th 2021, Patrick gave an invited lecture in the online webinar series on the Molecular Bases of Proteinopathies hosted by the Ramamoorthy lab at the University of Michigan. Patrick talked about our research on polyglutamine protein aggregation and the structural biology of Huntington’s disease.The seminar title was “The Structural Biology of Protein Misfolding in Huntington’s disease”. If you are interested, the seminar has been posted to YouTube, where it can be viewed here.