Congratulations to Raffaella for the publication of her second first-author paper, this time on the use of NMR spectroscopy to study how antibodies bind to the aggregated state of huntingtin protein fragments, relevant to Huntington’s disease. This work is part of our ongoing research on the biochemistry of Huntington’s disease, and was supported by funds from the European Huntington Disease Network EHDN and the CampagneTeam Huntington.
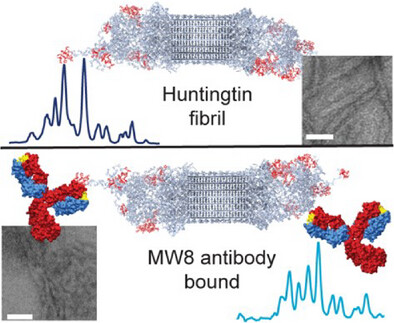
Antibodies are widely used as diagnostic tools in HD research, and also are considered as potential treatment modalities. An example of the latter approach can be found in the case of Alzheimer’s disease, where monoclonal antibodies are approved for clinical use. A common use of antibodies in HD research is to use them to identify and locate different HTT fragments, in soluble but especially also aggregated forms. Like other amyloid disease proteins, the aggregation of HTT (fragments) leads to different types of aggregates. In such ‘polymorphic’ protein aggregates, you can see differences in the buried core structure, but also changes in the protein parts that are exposed on the surface of the protein deposits. This explains how one can use antibodies against different epitopes to distinguish or identify different types of protein aggregates, as has been widely done in HD research, including in pivotal studies of HD patient materials.
However, it is difficult to really study how these antibodies bind the protein aggregates, due to their fibrillar nature and the propensity for antibodies to make samples even more heterogeneous. Here, we showed how solid-state NMR methods can be used to study these interactions – observing how the widely used MW8 antibody binds to its epitopes even in the aggregated protein samples. We observed changes in the dynamics of the epitope, but also higher-order effects on the clustering or clumping of protein fibrils. These types of changes have potential relevance to the biological properties of the aggregates, potentially offering a means of reducing their toxic (or stress-inducing) impacts on (neuronal) cells.
For more information, see the paper, available at the journal. Note that our paper is also included in the journal’s ‘Hot Topic’ collection about Amyloids.
Full citation:
Raffaella Parlato, Greeshma Jain, Alessia Lasorsa, Patrick C.A. van der Wel (2025) Probing How Anti-huntingtin Antibodies Bind the Fibrillar Fuzzy Coat Using Solid-State NMR. Chemistry – a European Journal, e01676 (in press) DOI: https://doi.org/10.1002/chem.202501676
(The corresponding bioRxiv preprint and a Zenodo data repository are also available online)