Tag: ssnmr
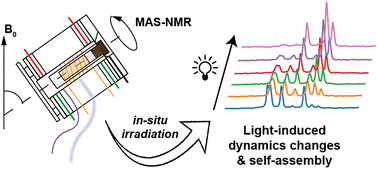
Publication: In-situ irradiated MAS NMR to probe dynamic transitions.
We are excited to report the publication of our paper on in-situ illumination during MAS NMR in the Journal of Materials Chemistry A. This publication is the culmination of a long project that found its origins quite a few years ago, even before the lab moved from Pittsburgh to Groningen. Photochemistry and optogenetics are increasingly influential topics in both chemical and biological research, respectively. The application of illumination and light triggers can be used to control chemical and biological processes. In this paper we focus on the intersection of such processes and materials science – demonstrating how magic-angle-spinning (MAS) NMR […]

New Publication: Silk-inspired hybrid materials – ssNMR and more.
Congratulations to PhD student Raffaella Parlato, as well as our collaborators from the group of Marleen Kamperman, with the publication of a new collaborative paper in the journal Communications Chemistry. Raffaella used solid-state NMR to study the structure and dynamics of (labeled) polypeptides within the hybrid polymer-peptide materials prepared by the Kamperman group. The employed peptides are poly-alanine peptides inspired by the structural motifs found in proteins in spider silk. Spider silk proteins use polyalanine segments to form stable higher order structures that enable the remarkable materials properties of silk. Here, these peptides were used to change the materials properties […]

Webinar: GlycoScience Webinar on polysaccharide ssNMR research
On 27 March Patrick gave an online presentation in the EGC webinar series, on the topic of using solid-state NMR (and isotope labeling) to study polysaccharides in various contexts. The title of the presentation was: “Labeling and solid-state NMR studies of hyaluronic acid and other polysaccharides: hydrogels, nanoparticles and more” EGC Webinar #86 Glycochemistry (March 27, 4 pm CET) Patrick van der Wel (Zernike Institute for Advanced Materials, University of Groningen, the Netherlands) “Labeling and solid-state NMR studies of hyaluronic acid and other polysaccharides: hydrogels, nanoparticles and more” A recording of the lecture is visible via YouTube. In the presentation […]

Publication: Review article on polyglutamine protein analysis by solid-state NMR spectroscopy.
Our new review article summarizing progress in polyglutamine protein studies by solid-state NMR is now available on the website of the journal Biochemical Society Transactions. This is an invited mini-review that was requested to cover the progress made in our understanding the aggregation and misfolding of mutant polyglutamine proteins implicated in diseases such as Huntington’s disease and several versions of spinocerebellar ataxia (SCA). We describe how studies from ssNMR has been used to better understand the structure of these protein aggregates, and thus helps us analyze how the aggregation process occurs and how aggregates may play a role in these […]

Publication: collaborative paper on perovskite-related materials, with Sn and Se ssNMR
Congrats to our collaborators from the group of Maria Loi and others for the publication of their new paper on in situ SnSe deposition as passivation for scalable and stable quasi-2D lead–tin perovskite solar cells, in the journal Energy & Environmental Science. Dr. Lasorsa in our group contributed tin and selenium ssNMR analysis to this interdisciplinary paper. For more information see the paper at the journal. Publication: Chen L, Tekelenburg EK, Gahlot K, Pitaro M, Xi J, Lasorsa A, et al. In situ SnSe deposition as passivation for scalable and stable quasi-2D lead–tin perovskite solar cells. Energy Environ Sci. 2023;10.1039.D3EE02507A.

Educational webinar on the use of ssNMR to measure dihedral angles.
Recently, Patrick gave an online lecture in the online webinar tutorial series of the Global NMR Discussion Meetings series (episode 70!). In this online lecture he introduced and discussed approaches to measure torsion angles (or dihedral angles) using advanced solid-state NMR spectroscopy. He discussed the basic idea of how these are implemented in ssNMR, but also how such structural data can be a useful complement to more traditional inter-atomic distance information obtained by ssNMR. You can now watch the recording of this lecture on Youtube, via this link. Want to learn even more? This lecture is connected to our recent […]

Publication: 13C labeling of hyaluronic acid polysaccharides (for ssNMR analysis)
Congrats to Pushpa and her collaborators on the new paper in the journal Carbohydrate Polymers. This new report describes Pushpa’s work to achieve the production of high-molecular-weight (HMW) hyaluronic acid, as part of her PhyCan (physics of cancer) project. Our interest in this polysaccharide stems from its important role in the extracellular matrix (ECM) of tissues, and in particular certain types of cancer tissues. In such tumors the amount of HA is upregulated, seemingly contributing to the progression of cancer development. A challenge in understanding this process is that HMW is difficult to study with most structural techniques, as it […]

Publication: SSNMR of alginate hydrogel (re)hydration
A new open-access publication by Mustapha and collaborators has been published in the journal Food Hydrocolloids, describing how he used various ssNMR measurements to probe alginate hydrogel structure and (re)hydration. Alginates can be cross-linked with calcium to form hydrogels, which are used in many different types of applications. This includes their use in slow-release drug delivery as well as food/nutritional applications. In this new paper, Mustapha shows how 1H, 2H and 13C ssNMR measurements can be used to see how these hydrogels are structured, but especially also how they are hydrated by the aqueous solvent. A key feature of the […]

Publication: Structural and motional changes in a cytochrome c – lipid complex implicated in apoptosis.
Congratulations to lab alum Dr. Mingyue Li and our collaborators on the publication of a new paper in the Journal of Molecular Biology. The paper is online via its DOI link. The paper describes how we used solid-state NMR spectroscopy to characterise the partial destabilization of the native fold of the protein cytochrome c, as it is bound to cardiolipin lipids. This protein-lipid complex is implicated in the process of programmed cell death, where it plays a key role in triggering the self-destruction of undesired or disease cells in higher organisms. Notably, the CL-bound protein catalyses the process of mitochondrial […]