Biomembranes are not just inert barriers, but represent a complex, dynamic and biologically important milieu, where structural biology and biophysical understanding go hand in hand. We study proteins associated with native-like membranes (i.e. fully hydrated lipid bilayers of complex composition), which are often critical for proper function and biological relevance.
We are studying the interactions of mitochondrial proteins with membranes, and in particular with the unusual mitochondrial phospholipid cardiolipin [1]. Binding to CL-rich membranes changes the function of cytochrome c, to turn it into a potent lipid peroxidase involved in mitochondrial apoptosis. In our NIH/NIGMS-funded research project we are using ssNMR and other techniques (including fluorescence-based assays) to determine the molecular mechanism of the pivotal early stages taking place in the mitochondria, in hopes of informing efforts to modulate these lethal processes.
Aside from such chemical changes in lipids, we also study how peptides and proteins disturb and disrupt the bilayer structure of membranes [3,4]. These processes play a critical role in potential cytotoxic mechanisms in neurodegeneration. At the same time, they are important for beneficial processes like the fusion and fission of biomembranes. Membrane curvature is increasingly recognized as an important regulatory mechanism, for signaling, trafficking and membrane targeting. For instance, caveolae are highly curved functional domains in the plasma membrane, enriched in both cholesterol and sphingomyelin. We have used ssNMR to study membrane-curving proteins, such as caveolin [3].
See also this old page, and the references below.
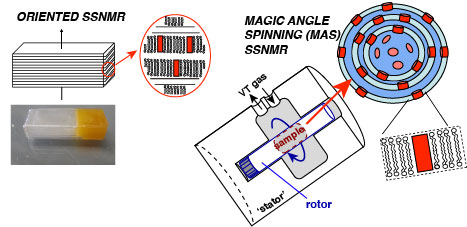
Selected References
- Mandal, A., Hoop, C.L., DeLucia, M., Kodali, R., Kagan, V., Ahn, J., Van der Wel, P.C.A.* (2015) Structural changes and pro-apoptotic peroxidase activity of cardiolipin-bound mitochondrial cytochrome c. Biophys. J. 109(9): 1873–1884
- Van der Wel, P.C.A.* (2014) Lipid Dynamics and Protein-Lipid Interactions in Integral Membrane Proteins: Insights from Solid-State NMR. eMagRes. (emrstm1356) [doi] [PDF Download]
- Eddy, M.T, Ong, T-C., Clark, L., Teijido, O., Van der Wel, P.C.A., Garces, R., Wagner, G., Rostovtseva, T.K., and Griffin, R.G. (2012) “Lipid Dynamics and Protein-Lipid Interactions in 2D Crystals Formed with the β-barrel Integral Membrane Protein VDAC1.” J. Am. Chem. Soc., 134: 6375 [doi]
- Hoop, C.L., Sivanandam, V.N., Kodali, R., Srnec, M.N., Van der Wel, P.C.A. (2012) “Structural Characterization Of The Caveolin Scaffolding Domain In Association With Cholesterol-Rich Membranes.” Biochemistry, 51: 90-99 [doi]
- Van der Wel, P.C.A. et al. (2000) “Tryptophan-anchored transmembrane peptides promote formation of nonlamellar phases in phosphatidylethanolamine model membranes in a mismatch-dependent manner.” Biochemistry 39: 3124-33. *
- Van der Wel, P.C.A. et al. (2002) “Geometry and intrinsic tilt of a tryptophan anchored membrane spanning peptide by 2H NMR.” Biophys. J. 83: 1479-1488 *
- Van der Wel, P.C.A. et al. (2007) “Orientation and motion of tryptophan interfacial anchors in membrane-spanning peptides.” Biochemistry 46: 7514-7524*